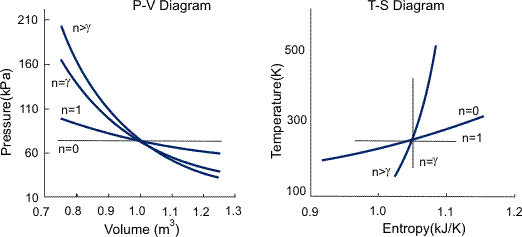
The term "polytropic" was originally coined to describe any reversible process on any open or closed system of gas or vapor which involves both heat and work transfer, such that a specified combination of properties were maintained constant throughout the process. In such a process, the expression relating the properties of the system throughout the process is called the polytropic path.
There are an infinite number of reversible polytropic paths between two given states; the most commonly used polytropic path is

where T is Temperature, S is Entropy, and C1 is a constant and is equal to zero for an adiabatic process. This path is equivalent to the assumption that the same amount of heat is transferred to the system in each equal temperature increment. In a reversible process following this polytropic path the heat and work transfer are as follows:

and

where H is Enthalpy.
Pressure and volume, and pressure and temperature are related by the following expressions:

and

where Cp and Cv are heat capacity at constant pressure and volume, respectively.
For an ideal gas this polytropic path simplifies to

where C2 and n are constants and n is called the polytropic index. The polytropic index characterizes the process, as summarized in Figure 1, and is given by

The equivalent pressure—temperature and work relationships are as follows:

and

Many gas compression and expansion processes may be usefully approximated by a polytropic process. In each case the polytropic coefficient must be determined experimentally by measurement of the heat and work transfer and the initial and final states.
Belt, Rowlinson, and Saville (1975) discuss the errors and inconsistencies which may arise when PVn = C2 is applied to nonideal gases and vapors.
REFERENCES
Bett, K. E., Rowlinson, J. S., and Saville, G. (1975) Thermodynamics for Chemical Engineers, The MIT Press, Cambridge MA.
References
- Bett, K. E., Rowlinson, J. S., and Saville, G. (1975) Thermodynamics for Chemical Engineers, The MIT Press, Cambridge MA.