One of the most important applications of thermodynamic processes is to do work. Work is any interaction between the system and its surrounding which can be used to lift a weight. The definition of work is
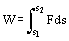
i.e., force multiplied by displacement in direction of force. The unit of work is 1 N m (Newton meter) = 1 J (Joule). Any force, such as the weight of a body, the force of a compressed spring or the attraction between two magnets can produce work. However, the force most commonly used is the pressure force, for example, in an internal combustion engine.

where p is the pressure, A the area, ds the distance moved and dV the change in volume.
If the fluid expands, V2 > V1, the piston moves upwards and work is done by the system. If the fluid is compressed, V2 < V1, the piston moves downward and work is done to the system. From this the definition of work follows: Work done by the system is positive. Work done to the system is negative.
In an open system, such as a steam turbine, the flow work during inlet and exit into the system contributes to the total work. The shaft work Ws which may be obtained by a rotating or reciprocating shaft is
