Gas chromatography (GC) is divided into two classes, gas liquid (partition) and gas solid (adsorption) termed GLC and GSC, respectively. GLC is subdivided into two modes, namely packed column, low performance (liquid stationary phase, SP, on a solid-inert support) and capillary or open tubular, column, high performance (liquid stationary phase, on the inner surface, physically adsorbed or chemically bonded) chromatography.
Of the two classes GLC is far more versatile than GSC, the latter only being usable for the separation and analysis of permanent, natural and synthetic gas mixtures and very volatile, gaseous organics, because of the adsorptive nature of the solid (sorbent) phase while the former can be applied to analyze organics over a wide range of volatilities (volatile solvents to C60 hydrocarbons).
In GLC applications, the capillary column mode has largely superseded the packed column mode, since the advent of fused silica open tubular (FSOT) columns in 1979 and the ability to apply and employ chemically-bonded SP (stationary phase) (GBC is a term that can be used to describe gas bonded-phase chromatography).
For all chromatographic separations, the solute is distributed (partitioned) between the liquid SP or solid sorbent and the mobile-gas phase (MP) and the equilibrium involved for GLC is:

where Kd is the distribution (equilibrium) coefficient, cL and cG are the molar concentrations in SP (L) and MP (G), respectively. The movement of the solute down the column under these conditions whereby Kd is directly related to cL/cG is termed linear chromatography. The ratio of the time spent by (or amount of) the solute in the two phases is given by k', the partition ratio (or capacity factor),


where β is the phase ratio, VG/VL.
GSC is governed by a process of adsorption involving an equilibrium between the solute in the solid sorbent phase and the gas phase.
The theory applied to packed and capillary columns stems from the 1950's, see, for example, Ettre (1965), and is required to account for
relative rates of migration solutes
rate of band broadening (BB) during migration.
The original theory applied was the Plate Theory whereby the movement of solute is considered as a series of stepwise transfers from one theoretical plate to the next (equilibrations of solute between SP and MP at each plate) which accounted for the variables influencing migration rates but not band broadening during migration.
The more complete treatment is provided by the Kinetic Theory using Plate Theory terminology which describes migration and band broadening quantitatively, and this theory is based on random mechanisms for the migration of solute molecules through a column.
The number of theoretical plates, N, and the height equivalent to a theoretical plate, H (in cm), are related to the length of a column, L (in cm), by
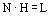
Using error theory and Gaussian distribution applied to the spread of velocities of particles about a mean velocity of the most average particle H is equated with the variance per unit length of the column

where σ2 the variance is the square of the standard deviation σ. An expression for N can be derived in terms of the chromatographic variables, retention time tt, and base width, wb, by eliminating L and σ.
or

where wb = 1.7w0.5.
The mechanisms of BB involve mass transfer processes in and out of the SP and in the MP during migration and are described in equations for packed and capillary columns by several terms which contribute to the overall H

where the A term accounts for the contribution of the packing (eddy diffusion), B is the coefficient for longitudinal diffusion important when the MP is a gas, Cs and Cm are the coefficients for resistance to mass transfer in and out of the SP and in the MP, respectively, and u is the mean average linear velocity of the MP. The equation reduces to three terms to describe the contributions to BB for capillary columns since A = 0 for no packing, and further to two terms B/ū+Cm·ū if the contribution of Cs·ū is negligible, i.e., when the film thickness of SP is a minimum, df = 0.1 μm and the column radius is small, rc = 0.2 mm.
Corrections can be applied to the equation to account for compression of the gas at the inlet (i) due to Pi > P ambient and decompression due to the pressure drop down the column Pi > P0 The pressure drop in the column is inversely proportional to the column diameter, dc, and thus wide bore columns will experience a lower ΔP than narrow bore columns.
For packed columns the packing and the limitation in dp (30 μm) serve to act as a barrier to flow (permeability); thus, the pressure drop is so severe that the effective column length is restricted to about 5 m, whereas capillary columns have a high permeability and columns of several orders of magnitude greater in length can be used.
The factors which influence the efficiency of a column measured in terms of H depend on ū, dp (particle size-packed), df (film thickness of SP), η (gas viscosity), rc and the column temperature, Tc. H decreases with a decrease in ū, dp, df, η and rc and with an increase in Tc
The parameters that can be determined directly from the chromatogram are the retention time (tr) and the base width (wb) or width at half height (w0.5). Using Equation (7) the number of theoretical plates, N, can be determined to give a measure of column efficiency dependent on k' for the solute analyzed. By obtaining tm for an unretained solute, the adjusted retention time t'r can be determined and the number of effective theoretical plates obtained by replacing tr by t'r in Equation (7).
The retention volume Vr can be determined by measuring the volumetric flow rate Fr at the column exit using the equation

The retention volume,  , can be corrected for the effect of gas compression and thus
, can be corrected for the effect of gas compression and thus

where j is the James-Martin compressibility factor, see James and Martin (1952).
The specific retention volume Vg is dependent only on the density of the SP, and the column temperature and has been extensively used to characterize solutes on columns with SP of widely differing polarities.
The resolution Rs of two solutes A and B (slower moving solute) is defined in terms of the retention times and base widths as follows

and complete separation is defined at Rs = 1.5. Rs is proportional to √N and N increases by a factor of √2 when the column length is doubled.
The selectivity, α, is a measure of the capacity of a column to separate two closely eluting solutes and is defined by the ratio

The difficulty in separation of two solutes A and B increases as a approaches unity.
The Retention Index (Kovats) was introduced to characterize different SP and solute functionality and is based on the retention parameters (t'r or k') of an homologous series, viz., n-alkanes on any SP for which I(x) is 100 × Cn Depending on solute functionality (nonpolar and polar) the k' of a solute will be shifted on a polar SP compared to a nonpolar SP and ΔI is a measure of the functionality—polarity, polarizability or bonding activity of the solute. The characterization of any SP is based on the polarity, P, obtained additively from the shifts in ΔI for selected solutes which are representative of the range of functionalities encountered (Rohrschneider and McReynolds constants).
For isothermal conditions, the relationship between log tr or k' and Cn (or 100 × Cn) is linear while for temperature programmed conditions, the relationship is directly linear (equal chromatographic spacing between successive components, cn and cn+1, of an homologous series, (e.g., n-alkanes or fatty acid methyl esters as examples).
With packed columns a very wide range of SP has been employed because of the limitations in the separation capability of packed columns. With the advent of fused silica (capillary) open tubular columns which are capable of high resolution the range of SP has narrowed dramatically such that only 5 or 6 phases (polydimethyl siloxane based or polyethylene glycol) are required to cover the entire range of solute functionality and volatility anticipated. As an example, a nonpolar polydimethyl siloxane SP of df 0.1 μm chemically-bonded to 25 m × 0.2 mm FSOT column will allow separation of a very wide range of solutes in a complex mixture according to their boiling points. Moreover, such columns are capable of chiral selectivity and thus can be used to separate enantiomers. Other features of SPs currently in use include high temperature stability and chirality for specialized applications.
For an introduction to the theory of chromatographic separations with reference to gas chromatography, see Bartle (1993).
REFERENCES
Bartle, K. D. (1993) Chapter 1, Gas Chromatography—a Practical Approach (Ed. P. J. Baugh), Oxford University Press.
Ettre, L. S. (1965) Open Tubular Columns in Gas Chromatography, Plenum Press, New York.
James, A. T. and Martin, A. J. P. (1952) Biochem. J. 50, 679.
Использованная литература
- Bartle, K. D. (1993) Chapter 1, Gas Chromatography—a Practical Approach (Ed. P. J. Baugh), Oxford University Press.
- Ettre, L. S. (1965) Open Tubular Columns in Gas Chromatography, Plenum Press, New York.
- James, A. T. and Martin, A. J. P. (1952) Biochem. J. 50, 679.
